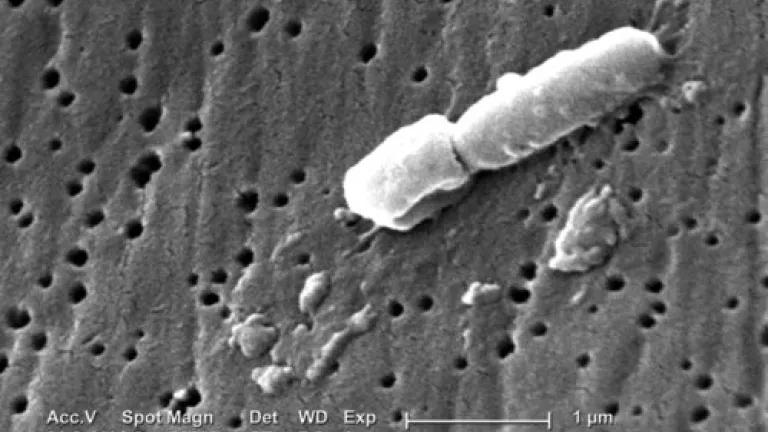
The latest news on antibiotic-resistant infections from the National Institutes of Health (NIH) is scary. That’s the only word that comes to mind when you hear about an antibiotic-resistant bacteria, or “superbug,” that survived careful and significant quarantine procedures; sickened 17 people, leading to six deaths; and which was unresponsive to all but one of the antibiotics available to treat it, and even that medicine did not work for four patients.
Colistin, the drug that doctors fell back on is an old drug with toxic side effects that can severely damage the kidneys, and that drug too is increasingly ineffective. Not only is our arsenal of antibiotics being rendered increasingly ineffective, there are few new antibiotics in the pipeline to replace the old drugs, and none to treat the kind of bacteria (gram-negative bacteria) that sickened the people at the NIH hospital, according to a doctor I spoke to recently.
These are the frightening realities of infections caused by antibiotic-resistant bacteria: longer illnesses, the use of stronger or more toxic medicines with greater side-effects, treatments that don’t work. And the Centers for Disease Control and Prevention (CDC) say that “[a]lmost every type of bacteria has become stronger and less responsive to antibiotic treatment when it is really needed.”
When leading medical and public health authorities, like the Institute of Medicine of the National Academies of Science and the Director General of the World Health Organization, are warning about the end of the effectiveness of antibiotics and of an era when commonplace infections could once again kill unless we make changes, it is a sobering prospect, and one that we need to heed. The CDC lists antibiotic resistance as one of its top concerns.
A world without effective antibiotics is a frightening prospect. We should be using every avenue possible to combat antibiotic-resistance, and getting rid of unnecessary uses of antibiotics that are causing resistance. That means reducing inappropriate use of antibiotics in hospitals and human medicine, but it also requires us to take the bull by the horns and address the massive and inappropriate use of antibiotics in livestock operations. That’s exactly what leading medical associations and health groups have urged, stating that “[o]veruse and misuse of important antibiotics in food animals must end, in order to protect human health.”
The CDC and hospitals already have significant efforts under way to reduce the use of antibiotics in hospitals and to contain resistant infections. Prescriptions for antibiotics in the US declined by 13% between 1997-98 and 2005-06. More needs to be done, but a lot is already being done. Now, we need to bring some of the same energy and effort to bear on antibiotic use in animal agriculture by bringing a halt to the unnecessary use of antibiotics for purposes other than treating sickness, i.e. for non-therapeutic purposes.
Only 20% of antibiotics in the United States are sold for use in humans, and the overwhelming majority of antibiotic use in the United States is on pigs, cows, chickens, and other poultry, comprising 80% of total use in the US, the vast majority for non-therapeutic purposes. Antibiotic use in livestock operations quadrupled between 1970 and 2010 from about 7 million pounds to about 29 million pounds. So, while we can improve antibiotic use in hospitals and the human medical setting, we should also be addressing antibiotic use in animal agriculture to combat this rising public health threat.
There’s a significant body of science that establishes that the use of antibiotics on livestock operations (and not just human use of antibiotics) contributes to the problem of antibiotic resistance in bacteria. The CDC has said that there is “strong scientific evidence of a link between antibiotic use in food animals and antibiotic resistance in humans,” including:
. . . multiple North American studies describing how:
• Use of antibiotics in animals results in resistant bacteria in food animals
• Resistant bacteria are present in the food supply and transmitted to humans
• Resistant bacteria result in adverse human health consequences (such as increased hospitalizations)
Once antibiotic-resistant bacteria proliferate on livestock operations, they don’t stay there. They hitch a ride on workers who work with the animals or with meat; they escape in soil, air, and water that comes into contact with animal waste and can then get on our vegetables and fish; they can be found on meat that makes its way into grocery stores; and bacteria can swap resistance genes with each other spreading resistance, including transfers from harmless bacteria to pathogenic bacteria (a new scientific study adds to the evidence on this swapping of genes). According to the CDC, “bacteria can become resistant to many [antibiotics] because of the transfer of one piece of DNA.” Meat in grocery stores is frequently contaminated with antibiotic-resistant bacteria, including superbugs resistant to 3 or more classes of antibiotics. People who acquire antibiotic-resistant bacteria can pass them on to other people and can even walk into hospitals carrying superbugs.
Antibiotic use at livestock operations has been described by prominent scientists as “a major driver of antimicrobial resistance worldwide,” it can lead to potentially disastrous consequences, and we should be moving at full speed to address that threat. The story of the NIH’s battle with one superbug infection is only the latest reminder of the urgency of the issue and the need to move to address it on all fronts as quickly as possible.
Yet, in the face of this rising public health threat, the Food and Drug Administration (FDA)—the agency that regulates the use of antibiotics in livestock—has been dawdling on this issue for decades. FDA needs to fast-track binding regulations to stop the non-therapeutic use of antibiotics in livestock-operations. We have called on them to do so and continue to push them to take meaningful action.
You can also send a powerful signal by asking the grocery stores and restaurants you frequent to sell meat raised without antibiotics and by choosing meat with the USDA Organic label or meat labeled “no antibiotics” backed by a “USDA Process Verified” or other independent certification.
Image of Klebsiella pneumoniae bacterium implicated in the NIH infections as seen on a scanning electron micrograph, courtesy of Centers for Disease Control and Prevention (CDC)

