Mercury Guide
Whether you're getting dental fillings or ordering sushi, keep these tips in mind to avoid exposure.

The coal-fired Mount Storm Generating Station in West Virginia
Eric Vance/EPA
Before scientists became aware of the toxic effects of mercury—it poisons the kidneys and nervous system—this seemingly magical metal was widely used in medicine, cosmetics, and industries like hatmaking. While mad hatters are a thing of the past, mercury exposure is now a serious and widespread health problem. Even small amounts of mercury can interfere with brain development, making exposure particularly risky for children younger than six and women in their childbearing years. The U.S. Environmental Protection Agency estimates—conservatively—that more than 75,000 babies are born each year with a greater risk of learning disabilities because of their mothers’ mercury exposure. NRDC puts that number as high as 265,000 newborns every year.
But enough of the scary stuff. Here are a few easy ways to minimize exposure.
Be finicky about fish.
Eating contaminated fish is the number one cause of mercury exposure in America. Mercury is spewed into the air from coal-burning power plants and factories. That pollution can travel halfway around the world and then settle into lakes, rivers, and oceans, where it is absorbed or ingested by small organisms and then starts working its way up the food chain, its concentration rising with each step. Big predatory fish, like sharks or tuna, can have especially high concentrations in their bodies.

Jarek Tuszyński via Wikimedia Commons, CC-BY-SA 4.0
You can’t see, smell, or taste mercury contamination in fish. Cooking has no effect on it, and you can’t avoid it by cutting off the skin or other parts of the fish. But you don’t need to eliminate fish altogether to stay in the mercury safe zone. Below are a few general rules of thumb. For more information about the specific mercury levels of your favorite fish, see our Smart Seafood Buying Guide.
- Avoid a few key species. King mackerel, marlin, orange roughy, shark, swordfish, tilefish, ahi tuna, and bigeye tuna all contain high levels of mercury. Women who are pregnant or nursing or who plan to become pregnant within a year should avoid eating these fish. So should children younger than six.
- Ease up on tuna. Tuna is the most common source of mercury exposure in the country. If you or your kids regularly eat canned tuna, stick to light or skipjack tuna, and limit it to less than two servings a week. A 130-pound woman can eat almost two six-ounce cans of light tuna a week and stay within the EPA-recommended safe zone for mercury. A four- or five-year-old child should eat only about four ounces of light tuna per week. The rules change when it comes to albacore tuna. Children should avoid that fish altogether, and women of childbearing age should stick to no more than four ounces per week.
- Make safer sushi choices. Popular sushi fish are often the apex predators of the food chain, so they tend to be high in mercury. If you’re pregnant, nursing, or planning a family, you can reduce mercury exposure from sushi by holding back on all types of tuna, mackerel, sea bass, and yellowtail. Fish and shellfish like eel, salmon, crab, and clam are lower in mercury.
- When in doubt, smaller is better. Forgo the big predators and pick the little guys, like anchovies, sardines, and scallops, which are lower in mercury.
Check your mercury level.
If you have concerns about your mercury consumption, ask your physician for a blood mercury test. Women with a high blood mercury level who are planning to start a family may decide to postpone pregnancy for a few months until that level drops.
Handle mercury thermometers with care. Or, better yet, replace them.
Although the liquid mercury in fever thermometers is less toxic than the form of mercury found in fish, inhaling it can still cause damage to the lungs, kidneys, and brain. In some cities, you may be able to exchange your old mercury thermometer for a free digital replacement. Household hazardous waste collection facilities and many pharmacies also collect and safely dispose of mercury thermometers.
If a mercury thermometer breaks, make sure that children do not touch the liquid mercury or inhale its vapors. Carefully sweep the little silver balls of mercury into a disposable plastic container and seal the container shut. Then take the container to your local household hazardous waste collection facility. Do not flush the mercury down the toilet, vacuum it, or throw it in the garbage can.
If the spill is on a rug or sofa, it’s best to get rid of the contaminated item. Mercury is extremely difficult to remove from carpets, upholstery, or other absorbent surfaces and will continue to vaporize over time.
Anatoliy Gleb/Adobestock
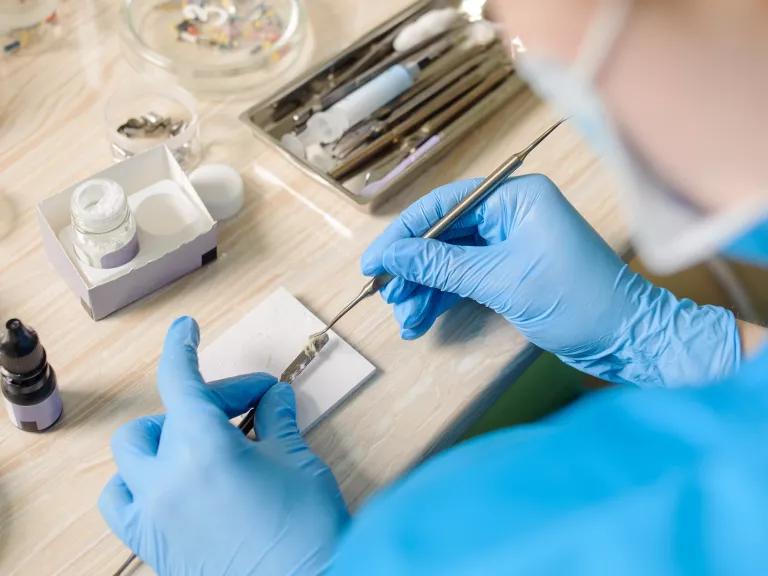
Leave your silver-colored dental fillings alone, and avoid them if you’re pregnant.
Silver-colored dental fillings contain as much as 50 percent mercury by weight and can release mercury vapor—especially when they are new or while you’re chewing. Pregnant women, or women planning to become pregnant within a few months, should avoid getting mercury fillings. Swallowing filling fragments poses less risk of harm because metallic mercury, unlike the mercury in fish, is not as easily absorbed in the stomach and intestines.
If you already have mercury fillings, most experts agree that it’s best to leave them in place because mercury can vaporize and be inhaled during the removal process. The dentist’s drill can also release small bits of the filling, which can get embedded in cheeks and gums.
The most popular alternative to mercury fillings is composite, or porcelain, fillings. However, these fillings contain the hormone-disrupting chemical bisphenol A, or BPA. Gold fillings appear to be the safest alternative but are considerably more expensive. Discuss the benefits and drawbacks of each option with your dentist.
Don't stress about most vaccines.
In late 1990s, NRDC and others successfully pressed for the removal of thimerosal, a preservative that contains ethyl mercury, from childhood vaccines to limit mercury exposure. The flu vaccine is the only routine childhood vaccine that still sometimes includes thimerosal, but parents can request a mercury-free version. Some of the combined diphtheria and tetanus vaccines may contain trace residues of thimerosal, but the amounts are extremely small.
This NRDC.org story is available for online republication by news media outlets or nonprofits under these conditions: The writer(s) must be credited with a byline; you must note prominently that the story was originally published by NRDC.org and link to the original; the story cannot be edited (beyond simple things such as grammar); you can’t resell the story in any form or grant republishing rights to other outlets; you can’t republish our material wholesale or automatically—you need to select stories individually; you can’t republish the photos or graphics on our site without specific permission; you should drop us a note to let us know when you’ve used one of our stories.

“Forever Chemicals” Called PFAS Show Up in Your Food, Clothes, and Home
The Particulars of PM 2.5
Mercury’s Journey from Coal-Burning Power Plants to Your Plate