And I Thought They Were God's Sneezes...
A new study shows where clouds come from—and what that means for climate research.

The earth’s atmosphere contains more than 3,000 cubic miles of water, slightly more than the volume of Lake Superior. And all that H2O has complicated effects on the planet’s temperature.
On one hand, water vapor is a powerful greenhouse gas, because it absorbs some of the heat trying to escape the atmosphere. On the other hand, water vapor is the primary ingredient in clouds, which can block incoming solar radiation and help to cool the planet. Understanding what transforms water vapor into clouds is therefore critical to building a reliable model of the future climate. A new study, released today in the journal Science Advances, takes a big step in that direction.
If you just want the basics on cloud formation, Google has you covered. Clouds form when invisible water vapor condenses onto airborne particles, aggregating into visible masses of water droplets. But you can go a lot deeper than that, especially when you look into the nature of those airborne particles, which helps dictate the traits and behavior of clouds.
Scientists call these tiny airborne particles “aerosols.” When water droplets condense on them, that process is called nucleation. (Nucleation isn’t unique to water vapor. You may notice that the bubbles in your beer or soda always seem to form in the same spots—they’re nucleating on contaminants or imperfections in your glass.) Many different substances in the atmosphere can serve as substrates for the nucleation of water vapor, but they can be roughly separated into two categories: natural and manmade (i.e. air pollution).
Scientists who study clouds sometimes point out that no one really knows what the sky looked like 200 or more years ago, because the Industrial Revolution vastly increased the number of particles onto which atmospheric water vapor could nucleate. Soot from diesel fuel and fireplaces, particulate matter from coal-burning power plants, and the products of countless other human activities all contribute to cloud formation. A paper published last year in the journal Science posited that modern skies are far cloudier than the blue skies of our ancestors, but it’s still an open question.
Preindustrial skies weren’t completely cloudless, of course, because of the many natural sources of aerosols. This is where today’s study comes in. The researchers reviewed satellite images of the world’s southernmost waters, where there is very little manmade pollution. What they found is that areas with the greatest concentration of water droplets also happened to be the same places where the green from the sea’s phytoplankton shone brightest. The connection between the two was obvious.
Marine phytoplankton contribute to cloud formation in two ways. First, they produce dimethylsulfide, or DMS. You’re familiar with it, even if you don’t know it, because DMS is the primary component of that sea smell you pick up near the ocean. After some chemical changes, the predecessors to DMS are useful nucleation sites for water vapor. The second phytoplankton contribution comes from sea spray. When the ocean froths and bubbles, it ejects tiny bits of organic matter that come from phytoplankton. Those particles can also help water vapor to condense.
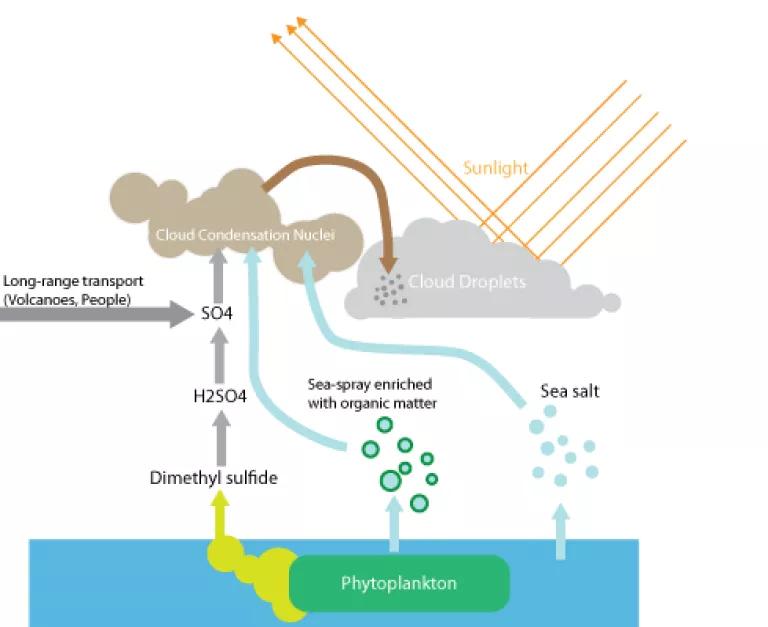
The researchers found that these particles have a huge effect on the reflectivity of the clouds over the southern ocean.
“In the summer, we get about double the concentration of cloud droplets as we would if it were a biologically dead ocean,” says Daniel McCoy of the University of Washington, one of the study’s lead authors.
The implications for climate models are significant, because clouds account for a large portion of the variation in our predictions.
“Unfortunately, the climate system is very sensitive to little changes in the cloud,” Andreas Muhlbauer, a research scientist at the Joint Institute for the Study of the Atmosphere and Ocean at the University of Washington, told Scientific American last year. “So being off by just a bit in a climate model can have a significant impact on the ability to predict."
The new study should help climatologists refine their estimates. “We’d like to consider how changes in ocean acidification and sea-surface temperature affect the phytoplankton and emissions of dimethyl sulfide,” says Susannah Burrows, another of the study’s lead authors, based at the Pacific Northwest National Laboratory. “This source of organic aerosol is not represented in most climate models.”
Climate change research has gone from a fringe interest to arguably the world’s most important scientific pursuit in just a couple of decades. We’re now at the stage where scientists are chasing the last bits of uncertainty out of the system—the role of ocean currents, the impact of dark land replacing white snow, and the effects of clouds, among others. Studies about phytoplankton and water-vapor nucleation may not (at first) sound fascinating, but they are critically important. If not to you, then they will be to your grandchildren.
This article was originally published on onEarth, which is no longer in publication. onEarth was founded in 1979 as the Amicus Journal, an independent magazine of thought and opinion on the environment. All opinions expressed are those of the authors and do not necessarily reflect the policies or positions of NRDC. This article is available for online republication by news media outlets or nonprofits under these conditions: The writer(s) must be credited with a byline; you must note prominently that the article was originally published by NRDC.org and link to the original; the article cannot be edited (beyond simple things such grammar); you can’t resell the article in any form or grant republishing rights to other outlets; you can’t republish our material wholesale or automatically—you need to select articles individually; you can’t republish the photos or graphics on our site without specific permission; you should drop us a note to let us know when you’ve used one of our articles.
How to Become a Community Scientist
The Man Behind the Mission
The Clean Air Act 101