Less Mercury Pollution, Less Mercury in Bluefish. Hmm…
A weekly roundup of the best in science journalism, doodled.

Curbing pollution makes fish less poisonous. Win-win.
The levels of mercury found in bluefish in the Atlantic have fallen 43 percent since 1972. The authors of a new study, published Tuesday in the Environmental Science and Technology Journal, think this drop has a “tight correlation” with reductions in pollution from North American power plants over the same time period.
Emissions from coal-fired plants are the largest source of mercury in the atmosphere, and that mercury winds up in our waterways and oceans through rain. Mercury is bioaccumulative, which means its concentrations build up in the tissues of animals as the neurotoxin travels higher and higher up the food chain. Bluefish, which are large predators that migrate up and down the Atlantic from Maine to Florida, have some of the highest amounts of mercury found in any fish. Like other top predators such as tuna, they are considered the primary source of mercury in humans. Among U.S. coastal areas, women in the East have the highest levels of mercury in their blood.
Although a move to further limit mercury emissions under the Clean Air Act was nixed by the Supreme Court last month, efforts over the decades to reduce the toxins coming out of smokestacks have had an impact. A report last year looking at the top 100 U.S. electric-power producers found that mercury emissions fell 50 percent between 2000 and 2012. The researchers of this week’s study compared mercury levels in fish from 1972 with those caught in 2011 to see what difference the drop in emissions may have made in marine ecosystems.
“What the EPA decides clearly makes a difference as to how much mercury people consume,” Richard Barber, professor emeritus of biological oceanography at Duke University, told Environmental Health News.
It’s worth noting, however, that the seafood Americans consume comes from all over the globe, and mercury pollution knows no borders. Check out this seafood guide, compiled by NRDC (disclosure), to find some less toxic options.
* * *
Art imitates nature—this time, with iridescent paints.

A U.K.-based painter who has been fascinated with iridescence in nature since childhood has made it her life’s work to emulate the phenomenon. “I wanted to use the same technology as the fish,” Franziska Schenk told Nautilus. Clearly some creatures woke up like this, but it took this human artist a full year to figure out how to mix the right pigments into paints that could effectively mimic nature’s light show.
The colors we see are the result of pigments in objects that absorb certain wavelengths of light and reflect others. Our eyes pick up the reflected light waves and our brains process them as colors. But there are also “structural colors,” which is where iridescence comes in. These are the result of two surfaces reflecting light in different ways, and how those light wavelengths interact with each other. Imagine a thin, transparent film, like that on the skin of a cuttlefish (above). Some light reflects off the top surface, but some wavelengths penetrate the film and bounce back when they hit the film’s bottom surface. Eli Kintisch at Nautilus explains it well using a soap-bubble analogy:
“The beams reflected from the top and from the bottom then combine…In that combination, some wavelengths of light get amplified, while some get diminished, depending on the chemical nature of the film and its thickness. The colors that get amplified are the intense colors we see. The way the waves combine also depends on the angle from which our eyes view that light, which explains why iridescent colors shimmer and change when viewed from different angles.”
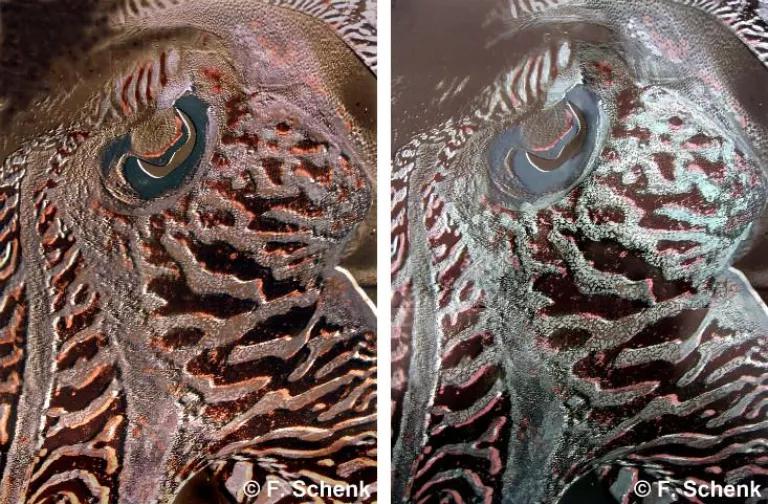
Now imagine trying to make paint do that. Schenk has had to relearn how to mix paint, this time using the rules of how light behaves, as opposed to pigment. The structure of her paintings now resemble that of a butterfly wing —she applies a bottom layer of pigment and then adds layers of reflectors to bounce light off the base layer, creating iridescent effects. Schenk has learned to paint in the same way that nature builds up color on its creatures.

* * *
In the southernmost seas, phytoplankton bring the clouds
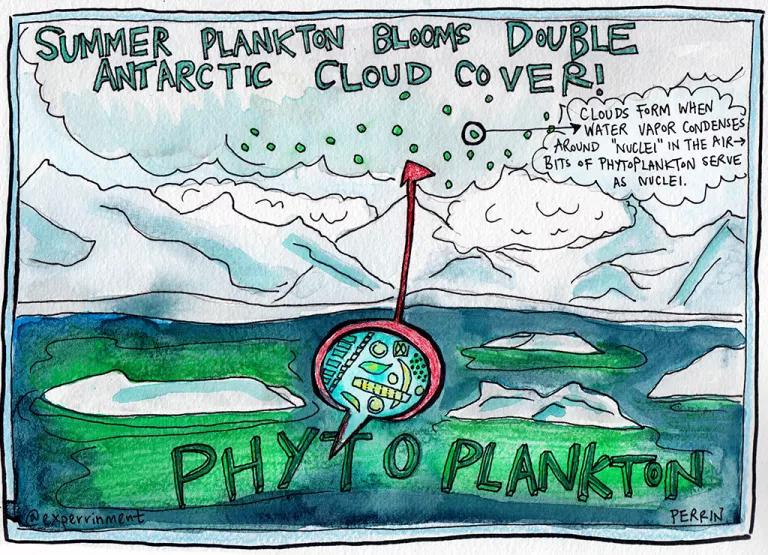
As you may have surmised, the Antarctic is far away from most human development. The distance from other land bodies is even so great that dust, pollen, and pollution don’t float down there as much. Those airborne particles help form clouds by acting as “nuclei” for water vapor to condense around. Given that the Southern Ocean is also the cloudiest area on earth, scientists thought it would be a great place to find out how clouds form naturally—without a lot of human-produced cloud “seeds” mucking up the data.
In a study published earlier this week, researchers found that summertime phytoplankton blooms almost double the cloud cover there. Phytoplankton bits, a.k.a. “natural aerosols,” enter the atmosphere through sea spray.
Because clouds affect how the earth’s atmosphere either traps or reflects heat from the sun, understanding cloud cover will help scientists build better climate models going forward.
* * *
Watch the best science-research videos of the year.
Everyone, a blog from the journal PlosOne, recently shared its favorite research videos of 2015 (so far). And the post is visual science on blast.
One captures the courtship process of java sparrows, which looks a lot like humans on a date at a concert—the birds twitch on opposite sides the cage for a while, come together in song, and then start making out.
Another shows the different methods macaques use to crush oysters. The names of these moves sound like crowdsurfing tactics at a metal concert. My favorite was “bimanual asymmetrical flip face hammering.”
And finally, the video I liked the best. This one features a trap-jaw ant using the mandible it typically grabs prey with to avoid becoming prey itself. Watch the ant escape the trap of its predator, the aptly named antlion.
This article was originally published on onEarth, which is no longer in publication. onEarth was founded in 1979 as the Amicus Journal, an independent magazine of thought and opinion on the environment. All opinions expressed are those of the authors and do not necessarily reflect the policies or positions of NRDC. This article is available for online republication by news media outlets or nonprofits under these conditions: The writer(s) must be credited with a byline; you must note prominently that the article was originally published by NRDC.org and link to the original; the article cannot be edited (beyond simple things such grammar); you can’t resell the article in any form or grant republishing rights to other outlets; you can’t republish our material wholesale or automatically—you need to select articles individually; you can’t republish the photos or graphics on our site without specific permission; you should drop us a note to let us know when you’ve used one of our articles.
How to Become a Community Scientist
How to Start Saving the Planet in 100 Days: the Joe Biden Edition
Why the EPA Needs Another William Ruckelshaus (1932–2019)