FDA’s Antibiotic Stewardship Plan Is Failing
A just-released FDA report shows that the agency’s approach to addressing the U.S livestock sector’s high-intensity use of medically important antibiotics is failing.

Livestock Rates of Use Persist Above 2017 Rates for Fourth Straight Year
A just-released FDA report, the latest in a series summarizing livestock antibiotic sales since 2009, shows that the agency’s approach to addressing the U.S livestock sector’s high-intensity use of medically important antibiotics is failing. Since 2017, at which point a veterinary directive was required to mix these antibiotics into animal feed and their use in healthy animals for the purpose of growth promotion had finally been made illegal, livestock producers have been using these precious medicines more intensively, not less.
Some of the crucial public health lessons from FDA reporting are often buried under spin. We are fortunate European officials over the last decade have transparently tracked livestock antibiotic sales and usage in up to 31 countries, and carefully documented their success in curbing antibiotic misuse and overuse. One direct result is that a proven policy roadmap exists for how U.S. farm antibiotic stewardship might get back on track.
Putting New FDA Data in Context
Since 2018, NRDC has compared, on an apples-to-apples basis, how intensively medically important antibiotics are used in livestock production in the U.S. and in Europe, where officials closely track patterns in antibiotic sales and usage to inform policy interventions. In early December 2022, we published our latest analysis, based on data from 2011 to 2020. Subsequently, we added an additional data point since 2021 data were released for Europe, as well as for the United States. Figure A portrays how, after a decade-long European campaign to improve veterinary antibiotic stewardship, antibiotic use rates for many of the region’s largest livestock-producing countries – including Germany, United Kingdom, France, Netherlands, and Denmark – are now two to six times less intense than in the United States.
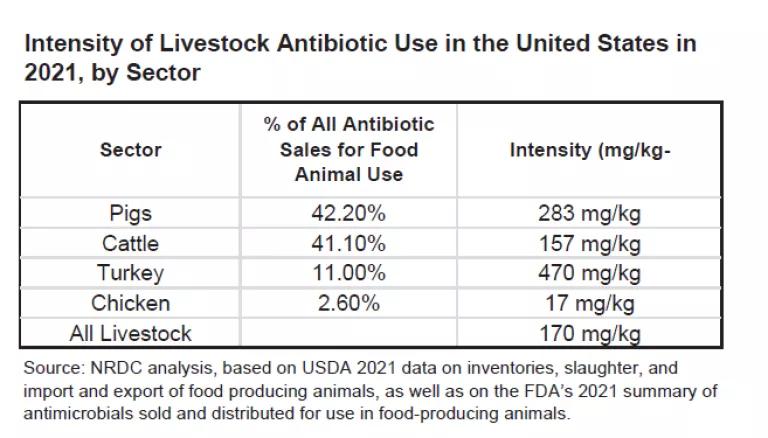
In fact, Europe now raises 75% more livestock by total weight than does the US on a fraction of the land area, and with less than half the rate of antibiotic use. The intensity of livestock antibiotic use, aggregated across all 31 European countries reporting data, is just 84 mg/kg compared to the 170 mg/kg intensity of use we attribute to the U.S. livestock sector, based on the just-published sales data.
Four years after the FDA banned further use of medically important antibiotics mixed into animal feed for growth promotion, the overall intensity of livestock antibiotic use is higher, not lower. That policy step hasn’t resulted in meaningful, lasting antibiotic stewardship. Europe also banned the last antibiotics for growth promotion in 2006. Since 2011, however, producers have found a way to reduce the intensity of antibiotic use across Europe by an additional 47%.
The lack of equivalent stewardship gains in the U.S. is mostly attributable to the U.S. pork industry, which in 2021 accounted for more than 42% of the nearly 6 million kilograms of livestock antibiotics sold for use. According to NRDC’s analysis, the industry also used these precious medicines 18% more intensively in 2021 than it did in 2017. By comparison, antibiotic intensity of use was also up by 10% in turkey and 3% in U.S. cattle production, but dropped 43% in chicken over the same time span.
Policy Implications: There’s A Better Way Forward
A recent report called out the FDA’s “leaderless dysfunction” when it comes to needed policy reforms to address public health risks related to food. Extensive overuse of medically important antibiotics on U.S. livestock farms is no exception. With the FDA's lack of action to date, we must look to Congress and the White House to get livestock antibiotic stewardship back on track while continuing to press FDA to lead.
The wheel doesn't need to be reinvented. Europe offers more than a decade worth of analysis and policy innovation, leading to proven success. As we have for several years, we urge that U.S. policymakers must adopt more of the core approaches that have already worked elsewhere:
- FDA (on its own or at the behest of Congress) needs to establish a national target to curb livestock antibiotic use by 50% by 2025 (relative to a 2010 baseline). The U.S. National Action Plan rests on CDC targets to curb antibiotic use and overuse in human settings; we are long overdue for similar targets and timelines to curb avoidable antibiotic use in livestock.
- Clear targets give focus to national efforts to reduce antibiotic use. But targets alone are meaningless if there’s no collection of data to measure progress towards meeting those targets. Roughly two-thirds of all medically important antibiotics sold for U.S. livestock are mixed into animal feeds at feed mills; under existing FDA requirements, the mills retain important information about this antibiotic use and the associated animal feeds. However, the FDA has never collected these data. FDA already collects user fees from the drugs’ makers; we urge Congress to make sure FDA uses these fees to collect and analyze the feed mill data, so that possible patterns of antibiotic misuse can finally be identified and addressed.
- At the outset of 2022, Europe carried out a third critical policy reform when it effectively banned future use of feed antibiotics for so-called disease prevention purposes. My next blog will talk more about why preventive antibiotics are unwise and unnecessary.


